Parts are the basic constructing blocks of matter. They’re the smallest items that can’t be damaged down into easier substances via chemical processes. All the pieces round us—air, water, soil, and dwelling organisms—is made up of mixtures of those parts. Though scientists have recognized 118 identified parts, solely a fraction are naturally occurring, and even fewer are important for all times.
Understanding how parts are organized, how they behave, and the way they contribute to dwelling programs is important to fields comparable to chemistry, biology, drugs, and environmental science. On the coronary heart of this understanding lies the periodic desk, a strong instrument that reveals the bodily and chemical properties of each identified component.
What Are Parts and Why Are They Essential?
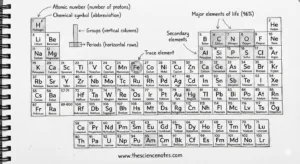
A component is a pure substance consisting of just one kind of atom. Every component is outlined by its atomic quantity, which represents the variety of protons in its nucleus. This quantity determines a component’s identification and its place on the periodic desk.
Whereas there are 118 identified parts, solely 92 happen naturally on Earth. Much more placing is the truth that people require simply 25 parts to reside and reproduce. Of these, solely 4 parts—oxygen, carbon, hydrogen, and nitrogen—make up roughly 96% of all dwelling matter.
These parts are important as a result of they kind the molecules that assist life, together with proteins, carbohydrates, lipids, and nucleic acids.
The Periodic Desk: Organizing the Parts
The periodic desk is a scientific association of all identified parts based mostly on growing atomic quantity and recurring chemical properties. Every sq. on the desk offers important data, together with:
Atomic quantity
Chemical image
Ingredient title
Atomic weight
For instance, sodium has the chemical image Na, derived from its Latin title natrium. This standardized system permits scientists all over the world to speak clearly and constantly.
Teams and Durations: Patterns in Chemical Conduct
Past atomic quantity, the periodic desk is organized into columns (teams) and rows (intervals) that replicate deeper chemical relationships.
Teams (Columns)
Parts in the identical group share comparable chemical properties, despite the fact that their atomic sizes could differ. This similarity arises as a result of they’ve the identical variety of electrons of their outermost shell, which largely determines how parts react and bond with others.
Durations (Rows)
Parts in the identical interval are extra comparable in measurement and have their electrons organized in comparable power ranges. Nonetheless, their chemical properties can fluctuate vastly throughout a interval because the variety of protons and electrons will increase.
This distinctive group permits scientists to foretell how parts will behave, even when they’ve by no means been studied earlier than.
Main Parts That Make Up the Human Physique
All dwelling organisms on Earth include oxygen, carbon, hydrogen, and nitrogen. In people, these 4 parts account for 96% of whole physique mass.
Oxygen (O): Important for mobile respiration and power manufacturing
Carbon (C): The spine of all natural molecules
Hydrogen (H): Performs a key position in power switch and molecular construction
Nitrogen (N): Essential for proteins and nucleic acids like DNA
These parts kind advanced molecules that drive each organic course of, from metabolism to copy.
Secondary Parts in Residing Matter
The remaining 4% of the human physique consists primarily of the next parts, listed so as of abundance:
Calcium (Ca): Helps bones, tooth, and muscle operate
Phosphorus (P): Important for DNA, RNA, and power switch (ATP)
Potassium (Ok): Regulates nerve alerts and fluid stability
Sulfur (S): Present in sure amino acids and proteins
Sodium (Na): Helps keep fluid stability and nerve impulses
Chlorine (Cl): Essential for digestion and electrolyte stability
Magnesium (Mg): Required for enzyme operate and muscle exercise
Although current in smaller quantities, these parts are important for sustaining regular physiological features.
Hint Parts: Small Quantities, Massive Influence
Hint parts are parts required by the physique in extraordinarily small portions—lower than 0.01% of whole physique weight—but they’re important for well being and survival.
Iron: A Essential Hint Ingredient
One of the vital vital hint parts is iron (Fe). Iron performs a central position in pink blood cells by serving to hemoglobin bind oxygen and transport it all through the physique.
A deficiency in iron can result in iron-deficiency anemia, a situation marked by:
Fatigue
Shortness of breath
Weak spot
Irregular coronary heart rhythms
This highlights how even minute quantities of sure parts can have profound results on human well being.
Dangerous Parts and Their Results on Residing Organisms
Not all parts are helpful. Some may be poisonous and even deadly, significantly heavy metals.
Mercury and Heavy Steel Toxicity
Mercury is a heavy metallic that may trigger extreme well being issues even at low ranges of publicity. Relying on the tissues affected, mercury toxicity can harm the nervous system, kidneys, and different organs. In bigger doses, it may be deadly.
One of many main issues with heavy metals is their capability to build up in dwelling tissues over time, a course of referred to as bioaccumulation.
Environmental Influence and Bioremediation
Heavy metals usually enter ecosystems via industrial air pollution and might transfer via the meals net, starting with main producers like vegetation and algae and ultimately affecting larger trophic ranges, together with people.
Scientists are exploring bioremediation, a organic strategy to eradicating heavy metallic contaminants from the setting. This analysis requires a deep understanding of each:
Such efforts are essential for safeguarding ecosystems and human well being.
Conclusion
Parts are the muse of all matter and life itself. From the 4 main parts that dominate dwelling matter to hint parts that quietly assist important organic features, every performs a novel position. The periodic desk offers a strong framework for understanding these parts, their properties, and their interactions.
On the identical time, consciousness of dangerous parts like mercury underscores the significance of accountable environmental administration and ongoing scientific analysis. By finding out parts and their conduct, we acquire perception not solely into the character of matter but in addition into the fragile stability that sustains life on Earth.