Proteins are the molecular machines of life. Each cell in each dwelling organism relies on proteins to outlive, develop, talk, and reply to the atmosphere. From constructing tissues to catalyzing life-sustaining chemical reactions, proteins are important organic macromolecules.
On this detailed, student-friendly information, we are going to discover proteins, amino acids, peptide bonds, aspect chains (R teams), polypeptides, pH results, and protein folding in depth—making it ideally suited for highschool, undergraduate, and early medical science learners.
What Are Proteins?
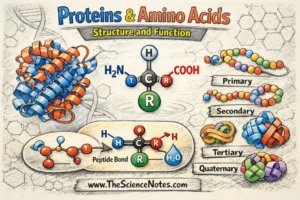
A protein is a protracted chain of amino acids joined collectively by covalent peptide bonds and folded right into a extremely particular three-dimensional (3D) construction. This 3D construction just isn’t random—it’s exactly organized, and it determines the protein’s organic operate.
Proteins are constructed from repeating items referred to as amino acid residues. When amino acids hyperlink collectively, they type a series often known as a polypeptide spine. The sequence of amino acids inside this spine comprises all the knowledge wanted for correct folding.
Relying on their size and structural complexity, amino acid chains are categorised as:
Oligopeptides (Peptides): Fewer than 20 amino acids
Polypeptides: Longer amino acid chains
Proteins: A number of folded polypeptides that perform particular organic capabilities
Not all polypeptides are useful proteins. A protein should undertake a steady 3D conformation to carry out its function successfully.
Amino Acids: The Basic Constructing Blocks
An amino acid is an natural molecule with a singular structural design. Every amino acid comprises:
A central alpha (α) carbon
A carboxyl group (–COOH)
An amino group (–NH₂)
A hydrogen atom
A variable aspect chain (R group)
The R group is what makes every amino acid chemically distinct.
The Significance of the R Group
The aspect chain (R group) determines:
Whether or not the amino acid is hydrophobic or hydrophilic
Whether or not it carries a optimistic, unfavourable, or impartial cost
Its potential to type hydrogen bonds
Its dimension and structural flexibility
For instance:
Glycine has the best aspect chain—a single hydrogen atom.
Proline has a inflexible ring construction that impacts protein folding.
Tryptophan has a big fragrant aspect chain.
How Many Amino Acids Are Utilized in Proteins?
Though lots of of amino acids exist in nature, solely 21 amino acids are used to construct proteins in eukaryotes (with 20 immediately encoded by the genetic code).
They’re represented utilizing:
Three-letter abbreviations: Gly, Val, Professional
One-letter abbreviations: G, V, P
The precise order of amino acids—often known as the first construction—determines the whole lot concerning the ultimate protein.
Peptide Bonds and Dehydration Synthesis
How Do Amino Acids Type Proteins?
Amino acids hyperlink collectively by way of a chemical response referred to as dehydration synthesis (also called a condensation response).
Throughout this course of:
The amino group (–NH₂) of 1 amino acid reacts with
The carboxyl group (–COOH) of one other amino acid
A molecule of water (H₂O) is launched
A powerful covalent peptide bond is shaped
This peptide bond is steady and types the spine of proteins.
Directionality: N-Terminus and C-Terminus
Polypeptides have directionality:
N-terminus: The start, with a free amino group (NH₃⁺)
C-terminus: The top, with a free carboxyl group (COO⁻)
Protein sequences are all the time written from N-terminus to C-terminus, reflecting how they’re synthesized inside cells.
Classification of Amino Acids by Aspect Chains
The chemical properties of aspect chains strongly affect protein folding and performance. Amino acids are generally grouped into 4 main classes:
1. Unfavorable Polar (Acidic) Amino Acids
Include a carboxyl group within the aspect chain
Carry a unfavourable cost at physiological pH
Instance: Aspartic acid, Glutamic acid
Take part in ionic interactions
2. Optimistic Polar (Fundamental) Amino Acids
Include amino teams within the aspect chain
Carry a optimistic cost at impartial pH
Typically work together with negatively charged molecules like DNA
3. Polar Uncharged Amino Acids
4. Nonpolar (Hydrophobic) Amino Acids
Lack charged or strongly polar teams
Repel water
Sometimes buried contained in the protein core
Vary from easy (glycine) to cumbersome (tryptophan)
Hydrophobic vs Hydrophilic Distribution
In aqueous environments:
This group stabilizes protein construction and is a serious driving power behind folding.
The Impact of pH on Amino Acid Chemistry
Amino acids are amphoteric, which means they will act as each acids and bases.
Their conduct relies on environmental pH.
At Low pH (Acidic Circumstances, ~pH 2)
At Excessive pH (Alkaline Circumstances, ~pH 13)
At Physiological pH (~7.4)
This twin cost permits amino acids to:
pH modifications can disrupt protein construction—a course of referred to as denaturation.
Polypeptide vs Protein: Understanding the Distinction
Though usually used interchangeably, there’s a distinction:
A polypeptide is a linear chain of amino acids.
A protein is a folded polypeptide (or a number of polypeptides) able to performing a organic operate.
Protein folding produces increased ranges of construction:
Major Construction: Amino acid sequence
Secondary Construction: Alpha-helices and beta-sheets
Tertiary Construction: 3D folding of a single chain
Quaternary Construction: A number of polypeptide subunits
Solely when correctly folded does a polypeptide develop into a useful protein.
Protein Dimension and Range
Proteins range tremendously in size and complexity.
For instance:
Thyroid-releasing hormone comprises 234 amino acids.
Connectin, an enormous elastic muscle protein, comprises over 34,000 amino acids.
This vary illustrates the extraordinary variety of protein structure.
Every protein is exclusive as a result of:
The variety of amino acids differs
The sequence of amino acids differs
The aspect chain interactions differ
The ultimate 3D folding sample differs
Even a single amino acid change can dramatically alter operate.
Why Proteins Are Important for Life
Proteins are one of many 4 basic organic macromolecules, together with:
Carbohydrates
Nucleic acids
Lipids
They carry out important roles in practically each organic course of:
Structural Assist
Motion
Catalysis
Transport
Immune Protection
With out proteins, mobile life could be not possible.
Key Studying Factors for College students
Proteins are composed of amino acids linked by peptide bonds.
The R group determines amino acid chemical conduct.
Peptide bonds type by way of dehydration synthesis.
The N-terminus and C-terminus outline protein directionality.
pH influences amino acid cost and protein stability.
Hydrophobic interactions drive protein folding.
Construction determines operate.