Liver organoids have grow to be an necessary device for finding out human liver biology, illness mechanisms, drug responses, irritation, and regenerative medication. Conventional two-dimensional liver cell cultures can present helpful data, however they typically fail to breed the complicated mobile communication that happens inside native liver tissue. The liver is just not product of hepatocytes alone. It comprises epithelial, endothelial, mesenchymal, and immune cell populations that work together repeatedly to manage metabolism, immune protection, harm response, and tissue restore.
Amongst these cell varieties, Kupffer cells are particularly necessary. Kupffer cells are liver-resident macrophages. They assist detect pathogens, clear particles, course of antigens, regulate irritation, and take part in liver restore and regeneration. Due to these features, they’re central to liver pathogenesis in situations akin to sepsis-related liver dysfunction, inflammatory liver harm, metabolic liver illness, fibrosis, and infection-associated hepatic stress.
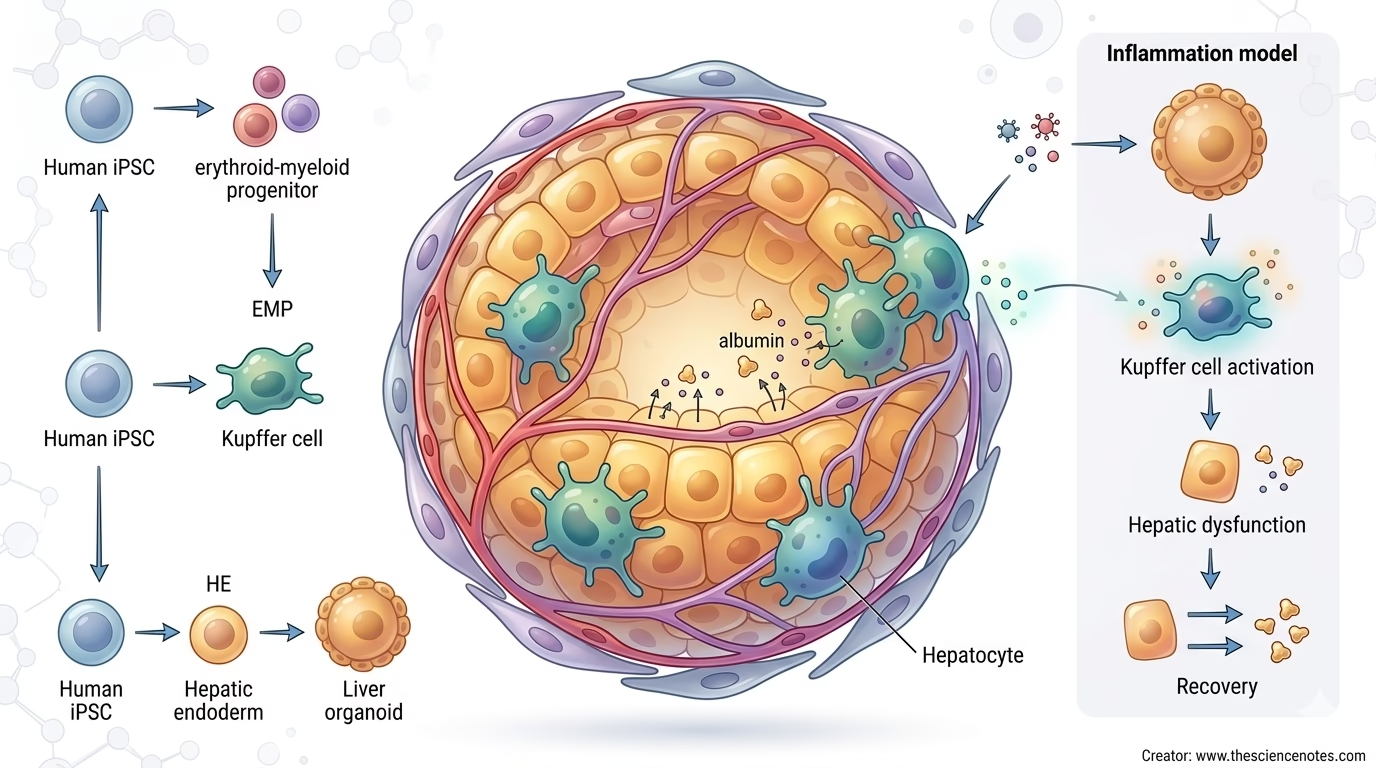
A latest STAR Protocols article by Yang Li, Yunzhong Nie, and Hideki Taniguchi describes a protocol for producing human induced pluripotent stem cell-derived liver organoids containing Kupffer cells, additionally referred to as KuLOs. The workflow integrates Kupffer cell progenitors into liver organoids by producing a number of cell lineages from human iPSCs after which combining them in a scaffold-free organoid co-culture system.
What Are Liver Organoids Containing Kupffer Cells?
Liver organoids containing Kupffer cells are three-dimensional liver-like constructions that embody each hepatic and immune elements. Within the protocol described by Li and colleagues, the organoids are generated from human iPSCs and comprise a number of iPSC-derived lineages:
The ensuing organoids are known as KuLOs, that means Kupffer cell-containing liver organoids.
The important thing benefit of this mannequin is that it doesn’t deal with the liver as a purely epithelial organ. As an alternative, it incorporates tissue-resident macrophage biology into the organoid system. This makes the mannequin extra related for finding out liver irritation, immune-mediated hepatic dysfunction, and restoration after inflammatory harm.
Why Kupffer Cells Matter in Liver Organoid Fashions
Kupffer cells are among the many most considerable resident macrophage populations within the human physique. They’re strategically positioned within the liver to observe blood coming into from the portal vein and hepatic artery. This exposes them to vitamins, microbial merchandise, toxins, immune alerts, and circulating particles.
Their features embody:
Detecting inflammatory and microbial signalsClearing apoptotic cells and broken materialProducing cytokines and chemokinesSupporting liver restore and regenerationParticipating in iron dealing with and antigen processingModulating hepatocyte perform throughout inflammationContributing to liver harm when immune activation turns into extreme
As a result of Kupffer cells can both shield the liver or contribute to harm relying on context, together with them in organoid fashions is efficacious. A hepatocyte-only organoid could present metabolic exercise, but it surely can not totally mannequin immune-driven hepatic dysfunction. A Kupffer cell-containing organoid can higher symbolize how immune activation impacts liver perform.
That is significantly necessary for modeling sepsis-associated liver dysfunction, the place systemic irritation and endotoxin publicity can impair hepatic perform. Within the KuLO system, endotoxin stimulation can activate Kupffer cells, set off inflammatory cytokine launch, and scale back hepatic efficiency. This supplies a managed human-cell platform for finding out inflammation-related liver harm and restoration.
Why Use Human iPSCs for Producing KuLOs?
Human induced pluripotent stem cells are helpful as a result of they are often expanded and differentiated into many cell varieties. In organoid analysis, iPSCs present a versatile beginning materials for creating complicated, multicellular fashions.
Utilizing human iPSCs affords a number of benefits:
1. Human Organic Relevance
Animal fashions are helpful however don’t at all times reproduce human liver improvement, immune signaling, drug metabolism, or inflammatory responses. Human iPSC-derived fashions might help bridge this hole.
2. Multilineage Differentiation
The KuLO protocol makes use of iPSCs to generate a number of cell lineages wanted for organoid meeting, together with hepatic endoderm, endothelial cells, mesenchymal cells, and erythroid-myeloid progenitors.
3. Reproducibility and Scalability
iPSC-derived methods may be expanded and standardized extra successfully than many major human tissue methods, though line-to-line variability nonetheless must be managed.
4. Illness Modeling Potential
Affected person-specific or genetically edited iPSC traces could also be used to review inherited liver problems, immune dysregulation, inflammatory susceptibility, or disease-associated variants.
5. Drug Testing and Therapeutic Screening
As a result of KuLOs mix hepatic and immune components, they could help testing of compounds that modulate irritation, hepatocyte perform, macrophage activation, or liver restoration.
Overview of the KuLO Technology Technique
The protocol described by Li and colleagues follows a developmental logic. As an alternative of merely including mature macrophages to preformed organoids, it generates erythroid-myeloid progenitors, or EMPs, from human iPSCs and incorporates them into liver organoids throughout co-culture.
This issues as a result of Kupffer cells come up developmentally from early myeloid progenitor populations. By integrating EMPs into the organoid system, the protocol goals to imitate elements of tissue-resident macrophage improvement and integration.
The final workflow may be summarized in 4 main phases:
Put together and broaden high-quality human iPSCsDifferentiate iPSCs into EMPs and hepatic endodermCombine EMPs with hepatic, endothelial, and mesenchymal lineagesAllow organoids to mature and characterize Kupffer cell integration
The ultimate KuLOs comprise a number of cell varieties and may be evaluated for hepatic maturation, Kupffer cell differentiation, immune activation, and useful restoration after inflammatory problem.
Part 1: Making ready Human iPSCs for Differentiation
Profitable KuLO technology begins with high-quality human iPSC tradition. The protocol emphasizes that iPSCs must be wholesome, maintained underneath applicable tradition situations, and passaged earlier than differentiation.
This step is important as a result of poor iPSC high quality can scale back differentiation effectivity throughout a number of lineages. If iPSCs are burdened, overgrown, spontaneously differentiated, or saved at extreme passage numbers, downstream hepatic and hematopoietic differentiation could also be compromised.
Essential high quality concerns embody:
Sustaining iPSC colonies in an undifferentiated stateAvoiding spontaneous differentiation earlier than beginning the protocolUsing applicable extracellular matrix coatingControlling passage timingUsing iPSCs with preserved differentiation potentialCoordinating separate differentiation timelines for various cell lineages
The protocol notes that hepatic endoderm and EMPs shouldn’t be cryopreserved for co-culture on this workflow. As an alternative, they’re really helpful for contemporary use, whereas endothelial and mesenchymal progenitors could also be dealt with in keeping with beforehand established procedures.
This scheduling element issues. Organoid meeting relies on having a number of cell lineages prepared on the identical time.
Part 2: Producing Erythroid-Myeloid Progenitors from Human iPSCs
One of many central improvements of the protocol is the environment friendly technology of erythroid-myeloid progenitors, or EMPs. These progenitors are used because the supply of Kupffer-like cells within the organoid system.
EMPs are necessary as a result of they symbolize an early hematopoietic inhabitants with myeloid potential. Within the KuLO workflow, they’re differentiated from human iPSCs via a sequence designed to advertise hemogenic and myeloid lineage specification.
In the course of the differentiation course of, researchers monitor the emergence of related progenitor markers. The protocol highlights markers akin to:
CD43CD34CD115, also referred to as CSF-1R
The goal EMP inhabitants is described as CD43-positive, CD34-positive, and CD115-positive. These markers assist affirm that the differentiation course of is producing an appropriate Kupffer cell progenitor inhabitants.
Stream cytometry is used to look at differentiation effectivity. This is a crucial quality-control checkpoint as a result of the success of Kupffer cell integration relies on the standard and id of the EMP inhabitants.
If EMP differentiation is poor, the ultimate organoids could comprise fewer Kupffer-like cells or present inconsistent immune conduct. The protocol recommends monitoring differentiation rigorously and optimizing iPSC seeding density and reagent high quality when yields are low.
Part 3: Producing Hepatic Endoderm from Human iPSCs
The hepatic part of the organoid begins with differentiation of human iPSCs into hepatic endoderm, typically abbreviated as HE. Hepatic endoderm represents a developmental stage on the trail towards hepatocyte-like cells.
The HE differentiation course of consists of two broad phases:
Definitive endoderm specificationHepatic endoderm differentiation
This displays regular developmental biology. Throughout embryonic improvement, liver tissue arises from definitive endoderm, which then receives alerts that drive hepatic specification.
Within the KuLO protocol, profitable HE differentiation is assessed utilizing morphology, circulate cytometry, and gene expression evaluation. By the top of the HE differentiation section, the cells are anticipated to point out hepatic endoderm traits, together with polygonal morphology and expression of related markers.
Essential HE markers embody:
CD326, also referred to as EpCAMCD133HNF4AAFP
The protocol additionally notes that definitive endoderm markers akin to CXCR4 and CER1 are anticipated to point out transient expression throughout differentiation, whereas pluripotency genes akin to NANOG and POU5F1 ought to lower.
A powerful HE inhabitants is important as a result of it kinds the hepatic basis of the organoid. Poor HE differentiation can result in failure of organoid formation, diminished hepatic perform, or poor maturation.
Part 4: Co-Culturing A number of iPSC-Derived Lineages to Generate KuLOs
After producing the required cell varieties, the protocol combines them in a scaffold-free co-culture system. The organoids are shaped in U-bottom microwell plates, which assist cells self-aggregate into three-dimensional constructions.
The primary cell populations used for KuLO technology are:
iPSC-derived hepatic endodermiPSC-derived endothelial cellsiPSC-derived mesenchymal cellsiPSC-derived erythroid-myeloid progenitors
The protocol recommends a lineage ratio wherein hepatic endoderm is the dominant inhabitants, with endothelial, mesenchymal, and EMP populations included at decrease however significant proportions. The offered ratio is 5:1:1:1 for HE, ECs, MCs, and EMPs respectively.
This co-culture design helps organoid formation whereas enabling EMP-derived macrophage improvement in and across the organoids. Throughout co-culture, EMPs broaden and differentiate towards a Kupffer-like phenotype. In the meantime, the liver organoids bear useful maturation.
The mannequin is designed in order that organoids steadily develop hepatic options whereas additionally incorporating immune-cell performance.
What Are KuLOs Anticipated to Comprise?
KuLOs are multicellular constructions. They don’t seem to be composed of a single liver-like cell sort. Their worth comes from the interplay of a number of lineages.
Hepatic Cells
The hepatic part contributes liver-related features, together with albumin secretion, expression of hepatic markers, and metabolic exercise.
Endothelial Cells
Endothelial cells assist symbolize vascular-associated signaling. Though the protocol notes that the endothelial community stays restricted in contrast with native liver tissue, this cell sort provides necessary tissue complexity.
Mesenchymal Cells
Mesenchymal cells present stromal help and developmental cues. They could assist manage organoid structure and affect hepatic maturation.
Kupffer-Like Cells
The EMP-derived macrophage inhabitants differentiates towards a Kupffer-like phenotype, expressing markers akin to:
By roughly two weeks of co-culture, the protocol reviews that the majority EMP-derived cells can specific Kupffer-associated markers. These Kupffer-like cells contribute immune responsiveness and assist mannequin liver irritation.
Characterizing Kupffer Cells in Liver Organoids
Characterization is a serious a part of the KuLO protocol. Producing organoids is just not sufficient; researchers should affirm that the specified cell populations are current and useful.
A number of approaches are used.
Stream Cytometry
Stream cytometry helps analyze hematopoietic and Kupffer-like cell differentiation. The protocol recommends analyzing EMP-derived cells throughout co-culture and assessing Kupffer-associated markers akin to CD45, CD14, and CD163.
Stream cytometry may also assist distinguish different organoid cell populations utilizing lineage markers akin to:
CD326 for hepatic cellsCD31 for endothelial cellsCD45 for hematopoietic cells
Nonetheless, the protocol notes that dissociation of organoids could not completely symbolize the precise mobile composition as a result of some cell clusters could stay undissociated.
Entire-Mount Immunostaining
Entire-mount immunostaining permits researchers to visualise organoids in three dimensions. This could present the spatial group of hepatic cells and Kupffer-like cells inside or across the organoid constructions.
Markers akin to hepatic proteins and macrophage markers can be utilized to substantiate multilineage group.
Hepatic Operate Assays
The organoids may be assessed for liver-like perform via readouts akin to:
Albumin secretionHepatic marker gene expressionCYP3A4 enzyme activityAmmonia metabolism
Albumin secretion is particularly helpful as a result of it supplies a useful indicator of hepatic maturation over time. The protocol reviews that albumin secretion will increase throughout co-culture and stabilizes as KuLOs mature.
Gene Expression Evaluation
Gene expression can be utilized to evaluate hepatic maturation, Kupffer cell id, inflammatory activation, and lineage-specific improvement. That is significantly helpful for evaluating KuLOs with organoids missing Kupffer cells or with major liver tissue references.
Anticipated Outcomes of KuLO Technology
The anticipated final result is a useful human iPSC-derived liver organoid system containing Kupffer-like cells. These organoids embody progenitors and differentiated cells from a number of lineages and might mannequin immune-hepatic interactions.
By roughly day 14 of co-culture, KuLOs are anticipated to achieve a extra mature state. At this stage, hepatic features stabilize and EMP-derived cells present Kupffer cell marker expression.
Reported useful options embody:
Elevated albumin secretion over timeUpregulation of hepatic markers akin to ALB, A1AT, and CPS1CYP3A4 activityAmmonia metabolismDifferentiation of EMPs into Kupffer-like cellsEnrichment of Kupffer-related features akin to antigen processing and iron ion homeostasisResponsiveness to endotoxin-induced inflammatory stimulation
These outcomes make KuLOs a helpful platform for finding out how liver-resident macrophages affect hepatic perform throughout irritation.
Modeling Sepsis-Associated Liver Dysfunction
Some of the necessary purposes of KuLOs is modeling inflammation-associated liver dysfunction, together with situations associated to sepsis.
Sepsis is characterised by systemic irritation, immune dysregulation, and organ dysfunction. The liver is closely concerned as a result of it filters blood, responds to inflammatory mediators, produces acute-phase proteins, and regulates metabolism throughout systemic stress.
Within the KuLO mannequin, endotoxin stimulation can activate Kupffer-like cells. This could result in:
Kupffer cell activationPro-inflammatory cytokine releaseImpaired hepatic functionReduced liver organoid performanceRecovery after elimination of inflammatory stimuli
This makes KuLOs helpful for finding out each harm and restoration. Researchers can look at how inflammatory alerts disrupt hepatocyte-like perform and the way tissue perform could also be restored after inflammatory mediators are eliminated.
Any such mannequin could also be helpful for therapeutic screening. Compounds that scale back dangerous irritation or promote hepatic restoration may very well be evaluated in a human multicellular organoid context.
Analysis Purposes of Human iPSC-Derived KuLOs
1. Liver Irritation Analysis
KuLOs present a platform to review how Kupffer cells regulate inflammatory signaling inside liver-like tissue. That is helpful for exploring cytokine networks, macrophage activation states, and hepatocyte responses to immune stress.
2. Sepsis and Acute Liver Dysfunction
As a result of KuLOs can reply to endotoxin publicity, they’re helpful for modeling sepsis-like inflammatory liver dysfunction. Researchers can research how immune activation impairs hepatic perform and the way restoration happens.
3. Liver Regeneration and Restore
Kupffer cells take part in liver restore after harm. KuLOs could assist researchers examine macrophage-derived alerts that help or inhibit liver regeneration.
4. Drug-Induced Liver Harm
Many drug-induced liver harm fashions focus totally on hepatocytes. Including Kupffer-like cells could assist mannequin immune-mediated liver toxicity or inflammatory amplification of drug responses.
5. Metabolic Liver Illness
Continual liver ailments akin to fatty liver illness contain interactions between hepatocytes, macrophages, endothelial cells, and stromal cells. KuLOs could help mechanistic research of inflammatory-metabolic crosstalk.
6. Fibrosis Analysis
Though KuLOs are usually not a whole fibrosis mannequin, the inclusion of mesenchymal cells and Kupffer-like cells could help future adaptation for finding out fibrogenic signaling and macrophage-stromal interactions.
7. Customized Drugs
Affected person-specific iPSCs may theoretically be used to generate KuLOs for finding out particular person susceptibility to liver irritation, immune dysregulation, or genetic liver illness.
8. Therapeutic Screening
KuLOs could present a platform for testing anti-inflammatory brokers, hepatoprotective compounds, macrophage-modulating medicine, or regenerative therapies.
Why This Protocol Is Progressive
The primary innovation is the mixing of Kupffer cell progenitors into human iPSC-derived liver organoids via a developmental technique.
Many organoid methods lack resident immune cells. This limits their potential to mannequin irritation and immune-tissue communication. Different strategies could add macrophages externally, however these approaches could not totally replicate tissue-resident macrophage integration.
The KuLO protocol addresses this by producing EMPs from iPSCs and co-culturing them with hepatic and supportive lineages. This enables Kupffer-like cells to emerge inside a creating organoid atmosphere.
The result’s a extra complicated liver mannequin that higher displays the multicellular nature of human liver tissue.
High quality Management Issues
Dependable KuLO technology relies on a number of checkpoints.
iPSC High quality
Wholesome, undifferentiated iPSCs are important. Poor iPSC tradition high quality can scale back each hepatic and hematopoietic differentiation.
EMP Differentiation
The EMP inhabitants must be assessed utilizing markers akin to CD43, CD34, and CD115. Low EMP yield could scale back Kupffer cell formation.
HE Differentiation
Hepatic endoderm ought to present applicable morphology and marker expression. Low HE high quality can impair organoid formation and hepatic maturation.
Organoid Formation
Cells ought to combination into spheroid-like constructions after seeding into microwells. Failure to type organoids could point out poor HE viability, insufficient cell preparation, or lacking survival-supporting situations.
Kupffer Cell Marker Expression
Kupffer-like differentiation must be monitored utilizing markers akin to CD45, CD14, and CD163.
Hepatic Operate
Albumin secretion, CYP exercise, ammonia metabolism, and hepatic gene expression might help assess organoid maturation.
Limitations of KuLO Fashions
Though KuLOs are an necessary development, they aren’t good replicas of grownup human liver tissue.
Restricted Hepatocyte Maturity
The protocol notes that KuLOs present hepatic perform however stay much less mature than major human hepatocytes. This can be a widespread limitation of many iPSC-derived hepatic methods.
Incomplete Endothelial Community
The organoids comprise endothelial cells, however they don’t totally reproduce the complicated vascular structure of the liver. This may occasionally restrict purposes centered on sinusoidal biology or vascular irritation.
Organoid Variability
Organoid measurement, cell composition, and maturation could range throughout wells, iPSC traces, and differentiation batches.
Technical Complexity
The workflow requires coordination of a number of cell lineages with completely different differentiation timelines. This will increase experimental complexity in contrast with easier organoid fashions.
Partial Immune Illustration
KuLOs embody Kupffer-like macrophages, however the liver immune atmosphere additionally comprises different immune populations, akin to lymphocytes, neutrophils, dendritic cells, and circulating monocytes. These are usually not totally represented.
Troubleshooting Ideas
The protocol identifies a number of widespread issues and sensible causes.
Low EMP Differentiation Effectivity
Low EMP yield could consequence from poor iPSC high quality, suboptimal beginning density, or degraded cytokines and development components. The protocol recommends optimizing iPSC seeding density and utilizing contemporary or correctly saved dietary supplements.
Low HE Differentiation Effectivity
Poor HE differentiation can happen when iPSCs have diminished differentiation potential, when seeding density is just not optimum, or when development components lose exercise. Marker evaluation and morphology checks are necessary for diagnosing this drawback.
Failure of Organoid Formation
If cells stay scattered as a substitute of clustering, seemingly causes embody poor HE differentiation, low HE viability, or inadequate help throughout preliminary co-culture. The protocol emphasizes the significance of cell viability and applicable co-culture setup.
Organoid Loss Throughout Medium Adjustments
As a result of organoids are usually not bodily connected to the microwells, careless dealing with can disturb or aspirate them. Light and constant medium change is necessary.
Inconsistent Kupffer Cell Differentiation
This may occasionally replicate variability in EMP high quality, co-culture situations, or timing. Stream cytometry at a number of time factors might help observe differentiation dynamics.
Ceaselessly Requested Questions
What are KuLOs?
KuLOs are human iPSC-derived liver organoids containing Kupffer-like cells. They mix hepatic, endothelial, mesenchymal, and hematopoietic-derived immune elements in a three-dimensional organoid mannequin.
What are Kupffer cells?
Kupffer cells are liver-resident macrophages. They assist regulate irritation, clear particles, course of antigens, take part in iron metabolism, and help liver restore.
Why embody Kupffer cells in liver organoids?
Together with Kupffer cells permits organoids to mannequin immune-hepatic interactions. That is necessary for finding out liver irritation, sepsis-related dysfunction, drug-induced harm, and tissue restore.
What are EMPs?
EMPs are erythroid-myeloid progenitors. On this protocol, they’re generated from human iPSCs and used as Kupffer cell progenitors.
What markers are used to establish EMPs?
The protocol highlights CD43, CD34, and CD115 as necessary EMP-associated markers.
What markers are used to establish Kupffer-like cells?
Kupffer-like cells in KuLOs are characterised utilizing markers akin to CD45, CD14, and CD163.
What markers are used to guage hepatic endoderm?
Hepatic endoderm may be assessed utilizing markers akin to EpCAM/CD326, CD133, HNF4A, and AFP, together with downregulation of pluripotency markers.
How lengthy does KuLO maturation take?
The co-culture section takes about two weeks. Round day 14, KuLOs are anticipated to point out extra mature hepatic perform and differentiated Kupffer-like cells.
What liver features can KuLOs mannequin?
KuLOs can present albumin secretion, hepatic marker expression, CYP3A4 exercise, ammonia metabolism, and inflammatory response to endotoxin stimulation.
Are KuLOs equal to grownup human liver tissue?
No. KuLOs are superior in vitro fashions, however they continue to be much less mature and fewer structurally complicated than native grownup human liver.
Conclusion
Human iPSC-derived liver organoids containing Kupffer cells symbolize an necessary step towards extra sensible in vitro liver fashions. By integrating liver-resident macrophage-like cells right into a multicellular organoid system, KuLOs permit researchers to review immune-hepatic communication in a managed human-cell platform.
The protocol by Li, Nie, and Taniguchi focuses on producing erythroid-myeloid progenitors and hepatic endoderm from human iPSCs, then combining these cells with iPSC-derived endothelial and mesenchymal lineages to determine Kupffer cell-containing liver organoids. The ensuing organoids help hepatic maturation, Kupffer-like cell differentiation, inflammatory activation, and restoration after inflammatory problem.
For liver illness analysis, this mannequin is particularly helpful as a result of many liver situations contain communication between hepatocytes and immune cells. Sepsis-related liver dysfunction, inflammatory harm, drug toxicity, regeneration, and continual illness development all contain macrophage-hepatic interactions. KuLOs present a sensible analysis platform for finding out these processes in a human iPSC-derived system.
Though limitations stay, together with incomplete hepatocyte maturity and restricted endothelial community formation, KuLOs provide a extra complicated and biologically related mannequin than hepatocyte-only organoids. As organoid know-how advances, Kupffer cell-containing liver organoids could grow to be more and more helpful for illness modeling, drug screening, regenerative medication, and precision hepatology.
Supply Attribution
This text is predicated on the scientific content material offered from:
Li, Y., Nie, Y., & Taniguchi, H. “Protocol for producing liver organoids containing Kupffer cells utilizing human iPSCs.” STAR Protocols, Quantity 6, Situation 4, 2025.
Associated referenced work:
Li, Y. et al. “Integration of Kupffer cells into human iPSC-derived liver organoids for modeling liver dysfunction in sepsis.” Cell Studies, 2024.