Proteins are probably the most versatile and functionally various macromolecules within the organic world. Whereas DNA holds the blueprint for all times, proteins are the precise laborers that execute the directions. Nonetheless, a protein is not only a string of chemical elements; it’s a subtle molecular machine whose energy is derived solely from its form.
The method by which a linear chain of amino acids transforms into a fancy, three-dimensional masterpiece is called protein folding. Understanding this course of is prime to trendy biochemistry, because the “kind follows operate” rule dictates each breath we take, each beat of our coronary heart, and even how our our bodies battle off an infection.
On the most simple stage, proteins are polymers constructed from 20 completely different monomers referred to as amino acids. Every amino acid shares a standard core construction: a central carbon atom ($alpha$-carbon) bonded to a hydrogen atom, an amino group ($-NH_2$), a carboxyl group ($-COOH$), and a novel aspect chain often called the R-group.
The Synthesis of Polypeptides
Throughout the technique of translation within the ribosome, amino acids are joined collectively by way of peptide bonds. This covalent bond kinds by way of a dehydration synthesis response between the carboxyl terminus of 1 amino acid and the amino terminus of the subsequent.
The ensuing chain is known as a polypeptide. Whereas the phrases “polypeptide” and “protein” are sometimes used interchangeably in informal dialog, scientists distinguish them by their state: a polypeptide is the uncooked chemical chain, whereas a protein is a polypeptide that has folded into its purposeful, biologically energetic 3D conformation.
To handle the immense complexity of those molecules, scientists describe protein construction by way of 4 distinct hierarchical ranges.
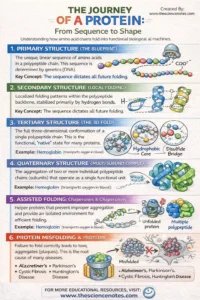
I. Main Construction: The Genetic Blueprint
The first construction is solely the linear sequence of amino acids. Regardless of its simplicity, this sequence is probably the most essential determinant of the protein’s future. The precise order of amino acids is dictated by the DNA sequence of the corresponding gene. As a result of every of the 20 amino acids has completely different chemical properties (dimension, cost, and hydrophobicity), their association determines precisely how the chain will ultimately appeal to or repel itself to kind a 3D form.
II. Secondary Construction: Localized Folding
Because the polypeptide emerges from the ribosome, it begins to kind localized “neighborhoods” of form. These are stabilized by hydrogen bonds between the atoms of the polypeptide spine (not the aspect chains).
Alpha-Helices ($alpha$-helices): A fragile, coil-like spiral held collectively by hydrogen bonds between each fourth amino acid.
Beta-Pleated Sheets ($beta$-sheets): Two or extra segments of the chain mendacity side-by-side, related by hydrogen bonds to kind a inflexible, sheet-like construction.
III. Tertiary Construction: The World 3D Fold
This stage represents the ultimate “native conformation” for many single-chain proteins. Whereas the secondary construction is concerning the spine, the tertiary construction is all concerning the R-group interactions. That is the place the protein collapses right into a globular or fibrous form primarily based on the chemistry of its aspect chains.
IV. Quaternary Construction: Multi-Unit Assemblies
A number of the most advanced proteins, resembling hemoglobin or DNA polymerase, encompass a number of polypeptide chains (subunits) that should come collectively to operate. This meeting is the quaternary construction. With out the right association of those subunits, the protein stays inactive.
3. The Forces That Drive Folding
Protein folding is a “search” for probably the most thermodynamically secure state. A number of key chemical forces act because the “engineers” of this course of:
The Hydrophobic Impact
That is maybe probably the most important power in protein folding. Within the watery setting of the cell, non-polar (hydrophobic) amino acid aspect chains naturally need to keep away from water. Because the protein folds, these hydrophobic residues cluster collectively within the inside “core” of the protein, whereas polar and charged (hydrophilic) residues stay on the outside to work together with water.
Molecular “Staples”: Disulfide Bonds
Cysteine is a novel amino acid as a result of its aspect chain accommodates a sulfur-containing thiol group ($-SH$). When two cysteines are introduced shut collectively throughout folding, they’ll kind a covalent disulfide bridge. These act like molecular staples, locking the protein into its ultimate, most secure form and defending it from being simply unfolded.
Van der Waals and Electrostatic Forces
Van der Waals Forces: As soon as the hydrophobic core is tightly packed, these weak points of interest between atoms present an additional layer of structural stability.
Ionic Bonds (Salt Bridges): Positively charged aspect chains (like Lysine) can appeal to negatively charged ones (like Aspartic Acid) to “zip” elements of the protein collectively.
For a very long time, scientists believed proteins folded solely on their very own (Anfinsen’s Dogma). Nonetheless, we now know that the mobile setting is just too crowded for many proteins to fold efficiently with out assist. Enter molecular chaperones.
Chaperonins: These are barrel-shaped protein complexes that act as “secure rooms.” An unfolded polypeptide enters the barrel, a “lid” closes, and the protein is allowed to fold in isolation, away from different molecules that may trigger it to clump or combination.
Warmth Shock Proteins (HSPs): These proteins improve in focus when the cell is pressured by warmth. They bind to uncovered hydrophobic areas of unfolding proteins to forestall them from sticking to one another and forming poisonous “clumps.”
The Position of Molecular Chaperones: The High quality Management Crew
For a very long time, it was believed that proteins folded solely on their very own primarily based solely on their sequence (Anfinsen’s Dogma). Nonetheless, the inside of a cell is a crowded, “salty soup” of organelles and different macromolecules. On this setting, newly synthesized polypeptides are at excessive threat of clumping collectively (aggregating) or folding into “dead-end” shapes that provide no organic utility.
To make sure survival, cells have developed a classy high quality management system led by molecular chaperones. These proteins don’t dictate the ultimate form of the protein—the amino acid sequence nonetheless does that—however they supply the help and setting needed for the protein to search out its “native conformation” effectively.
1. Chaperonins: The Isolation Chambers
Chaperonins, such because the well-studied GroEL/GroES advanced in micro organism, are barrel-shaped protein buildings. They act as “secure rooms” for folding.
Mechanism: An unfolded or partially folded polypeptide enters the central cavity of the “barrel.”
Isolation: A “lid” (chaperonin cap) closes the chamber. Inside this protected microenvironment, the protein is shielded from the crowded cytoplasm.
Folding: The setting contained in the barrel typically has chemical properties that favor right folding. As soon as the method is full, the lid opens, and the purposeful protein is launched.
2. Warmth Shock Proteins (HSPs): The Molecular Bodyguards
Warmth shock proteins, resembling Hsp70, are the cell’s first line of protection towards misfolding, particularly throughout environmental stress like excessive fever or pH modifications.
Mechanism: They determine and bind to uncovered hydrophobic areas on an unfolded polypeptide.
Prevention: By “masking” these sticky hydrophobic patches, HSPs forestall the polypeptide from sticking to different proteins within the cell.
Launch: Utilizing power from ATP, the HSP ultimately releases the protein, giving it one other likelihood to fold appropriately.
Comparability: Chaperonins vs. Warmth Shock Proteins
Whereas each are chaperones, they function at completely different phases of the protein’s life cycle.
FeatureHeat Shock Proteins (e.g., Hsp70)Chaperonins (e.g., GroEL/ES)Bodily FormSmall, clamp-like proteins.Giant, barrel-shaped complexes.Main MotionBinds to and stabilizes “sticky” areas.Gives an remoted “cage” for folding.TimingUsually acts early, whereas the protein is being made.Acts later, on partially folded intermediates.Vitality UseRequires ATP to bind/launch the protein.Requires ATP to shut the lid and cycle the barrel.PurposePrevents aggregation and “misfolding” throughout stress.Facilitates the ultimate 3D “native” fold.
Enzymatic Helpers: PDI and PPI
Along with chaperones, particular enzymes pace up the chemical “locking” of a protein:
Protein Disulfide Isomerase (PDI): This enzyme is essential for proteins that require disulfide bonds. It helps the protein quickly “take a look at” completely different bond combos till probably the most secure, right disulfide bridges are fashioned.
Peptidyl Prolyl Isomerase (PPI): This enzyme helps rotate bonds involving the amino acid Proline, which is usually a “kink” within the chain that may decelerate the folding course of.
With out this workforce of chaperones and enzymes, the “folding funnel”—the trail a protein takes to search out its secure form—could be too sluggish and susceptible to errors, resulting in the mobile “trash” that causes neurodegenerative ailments.
5. Architectural Variety: Globular vs. Fibrous
Proteins usually fall into two broad structural classes primarily based on their tertiary or quaternary shapes:
Globular Proteins
These are spherical, compact, and usually soluble in water. Their surfaces are coated in hydrophilic residues, making them good for transferring by way of the bloodstream or cytoplasm.
Examples: Hemoglobin (oxygen transport), Insulin (hormone signaling), and nearly all enzymes (catalysis).
Fibrous Proteins
These are lengthy, rope-like, and insoluble in water. They’re constructed for power and sturdiness quite than chemical reactivity.
Examples: Keratin (strengthening hair and pores and skin), Collagen (offering construction to tendons and bone), and Actin/Myosin (facilitating muscle motion).
6. When Folding Goes Unsuitable: Denaturation and Illness
Since a protein’s operate is only depending on its form, dropping that form—a course of referred to as denaturation—is normally catastrophic.
Causes of Denaturation
Warmth: Will increase kinetic power, vibrating the protein till weak hydrogen bonds break.
pH Modifications: Disrupts the ionic bonds (salt bridges) by altering the cost of the aspect chains.
Chemical compounds: Urea or detergents can disrupt the hydrophobic core.
Proteopathy: The Ailments of Misfolding
If a protein misfolds and the cell’s high quality management programs (like chaperones) fail to repair or destroy it, these proteins can combination into “amyloid plaques.” These plaques act like “molecular sand” within the gears of the cell, ultimately resulting in cell demise. That is the underlying mechanism for a lot of neurodegenerative circumstances:
Alzheimer’s Illness: Brought on by the buildup of beta-amyloid plaques.
Parkinson’s Illness: Linked to the misfolding of alpha-synuclein.
Cystic Fibrosis: Brought on by a single amino acid deletion that stops a membrane protein from folding appropriately, resulting in its destruction by the cell earlier than it will possibly ever operate.
Conclusion: The Precision of Organic Engineering
The journey of a protein from a easy genetic sequence to a purposeful 3D machine is likely one of the most outstanding feats of organic engineering. Each interplay—from the power of a covalent disulfide bond to the delicate “shyness” of a hydrophobic residue—is completely balanced to make sure the protein can carry out its life-sustaining function. As we proceed to map the “proteome,” our understanding of those folding pathways will unlock new remedies for ailments and permit us to design artificial proteins that would remedy international challenges in medication and trade.